Patient-20
Patient-20 had hip replacement surgery by Dr. Michael Bradford with Nevada Orthopedic & Spine Center, who implanted a previously used, defective, recalled device. It caused horrible pain and suffering, and the patient developed sepsis. Patient had to go back into the hospital, and a different surgeon removed the defective device.
A male patient had hip replacement surgery by Dr. Michael Bradford with Nevada Orthopedic & Spine Center in 2024, using a device manufactured by Exactech.
The patient was in horrible pain and suffering and also developed a serious case of sepsis. He had to go back into the hospital and found a good surgeon to help him, and the defective device was removed.
The new surgeon discovered that Dr. Bradford had used a previously used, defective, recalled orthotic hip replacement, produced by Exactech.
- In March 2023, the FDA warned about Exactech’s joint replacement devices made between 2004 and August 2021. The packaging caused the devices to degrade earlier than expected. Many people with faulty joint replacements require additional surgery.
- In July 2021, Exactech issued a worldwide Urgent Dear Healthcare Professional (DHCP) communication regarding Exactech Connexion GXL, moderately crosslinked polyethylene acetabular hip liners.
- On August 11, 2022, Exactech issued an updated and expanded recall for their hip replacement devices, noting an additional risk factor.
- In total, Exactech recalled more than 200,000 ankle, knee, and hip joint implants.
Notice the dates of the Exactech recall for hip replacement devices (July 2021 and August 2022) and the FDA warning (March 2023).
In 2024, Dr. Bradford used a previously used, defective, recalled device on this male patient, so Dr. Bradford used this device knowing full-well that it had been recalled.
The notification graphic on this page was found on Facebook.
Multiple lawsuits and class action lawsuits were filed against Exactech.
- People filed Exactech lawsuits after the Florida-based company recalled thousands of hip, knee, ankle and shoulder devices in 2021, 2022 and 2024 due to defective packaging. According to lawsuits, the defective packaging caused devices to fail early, leading to various complications, including the need for additional surgery.
- With over 143,000 recalled implants in question, patients are pursuing compensation for injuries like device failure and revision surgeries.
Exactech filed bankruptcy on October 29, 2024, in response to the mounting lawsuits. Ongoing litigation was paused as a result of the Chapter 11 filing.
As of October 2025, because of the Exactech bankruptcy case, no trials or settlements have yet occurred.
To Understand Modern Healthcare, Follow the Money
During COVID, hospitals lost significant revenues when elective surgeries were put on hold. To offset losses, many hospitals looked to the older adult population, performing record numbers of surgeries that were of little benefit to patients.
In addition to HMO payouts, hospitals stand to profit from Medicare and Medicaid, government programs designed to support aging Americans and the underprivileged. These programs are easy to exploit and are often the target of fraud. Unnecessary surgeries charged to such programs are a cash cow for unscrupulous practitioners.
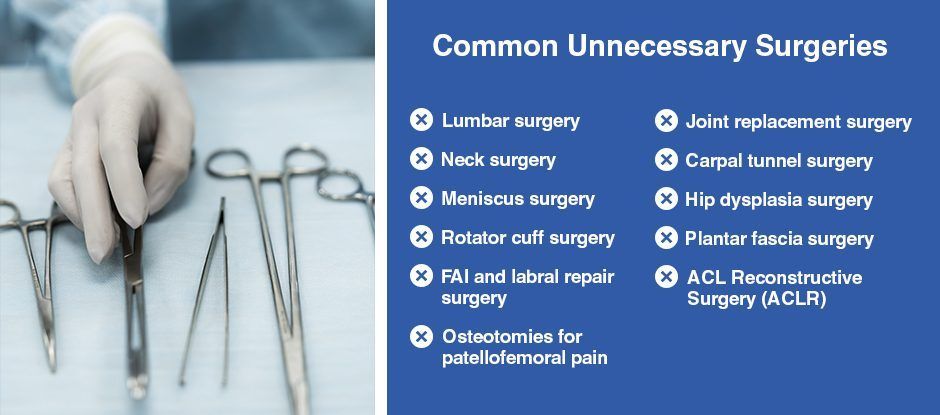
Common Unnecessary Surgeries
Many joint and back surgeries have little to no scientific evidence to support them, yet they are performed by the thousands each year. Some routine surgeries have been performed for decades with no better outcomes than physical therapy and regenerative treatment.
In many cases, the side effects of surgery are worse than the original condition. Sadly, surgeons tend to downplay negative side effects and often fail to disclose realistic recovery timelines.
Read the
article.
The notification graphic above was found on Facebook.
Dr. Michael Bradford used a defective, recalled device on a hip replacement patient, causing horrible pain and suffering and sepsis.
Patient found a new surgeon and had to undergo revision surgery to have the defective device removed.
Just say NO to Sunrise Hospital
and just say NO to Nevada Orthopedic
and just say NO to Dr. Michael Bradford

Patient injuries in defective, recalled implants manufactured by Exactech (used by Dr. Michael Bradford in Patient-20)
Unnecessary Surgeries
Joint and back surgeries are on the rise, not only in the United States but around the world. Yet surgical interventions are costly and pose substantial health risks, with no guarantee of successful outcomes.
In many cases, surgical outcomes are no better, and sometimes worse than, the outcomes of conservative care. Meanwhile, the cost of medical services has gone through the roof in an all-time low.
Gain insight into the issues plaguing mainstream medicine, the dramatic uptick in unnecessary procedures, and breakthrough holistic alternatives that deliver superior results.
It is impossible to explain the increase in unnecessary procedures without addressing the twin elephants in the room: The rising cost and diminishing quality of healthcare in the United States.
We can point to three primary factors driving these phenomena:
- The trend toward specialization, where practitioners zero in on single areas of human health while ignoring the integrative whole.
- A massive growth in healthcare administration, with administrators outnumbering doctors by a ratio of 10 to one.
- The advent of HMOs, where the tail of healthcare insurance protocols wags the dog of patient care.
Consider the following facts:
- In 2022, the average health insurance premium for a single individual in the US was $7,911 per year, and a whopping $22,463 per year for average family coverage. Meanwhile, the median income per US household is only around $70,000, making the average health insurance premium nearly 32% of median income.
- Most health insurance policies have minimum copay requirements amounting to thousands of dollars per household, in addition to other various out-of-pocket expenditures. In many cases, patient claims are denied by insurers, putting the patient between the rock of medical billing and the hard place of insurer denials. For many, bankruptcy is the only recourse to a system that puts patients last.
Today, compared to other wealthy nations, the US has the lowest life expectancy at birth, the highest death rates for avoidable conditions, the highest maternal and infant mortality, and the highest suicide rates.
Read the article.
